Thermo Fisher Scientific › Electron Microscopy › Electron Microscopes › 3D Visualization, Analysis and EM Software › Use Case Gallery
Microscopic organisms that penetrate calcareous structures by actively dissolving the carbonate matrix, namely microendoliths, have an important influence on the breakdown of marine carbonates.
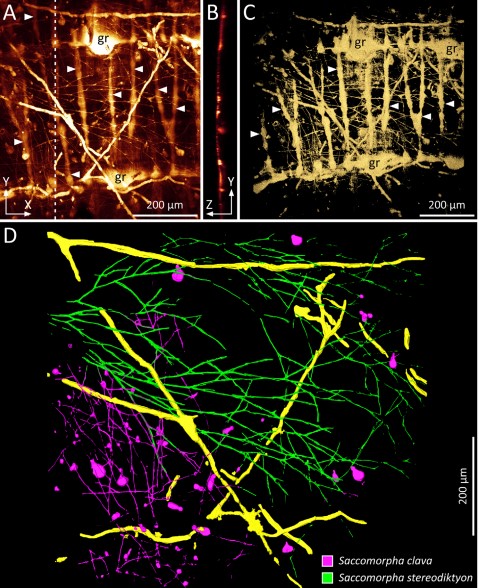
Microscopic organisms that penetrate calcareous structures by actively dissolving the carbonate matrix, namely microendoliths, have an important influence on the breakdown of marine carbonates. The study of these microorganisms and the bioerosion traces they produce is crucial for understanding the impact of their bioeroding activity on the carbonate recycling in environments under global climate change. Traditionally, either the extracted microendoliths were studied by conventional microscopy or their traces were investigated using scanning electron microscopy (SEM) of epoxy resin casts. A visualisation of the microendoliths in situ, that is within their complex microbioerosion structures, was previously limited to the laborious and time-consuming double-inclusion cast-embedding technique. Here, we assess the applicability of various fluorescence staining methods in combination with confocal laser scanning microscopy (CLSM) for the study of fungal microendoliths in situ in partly translucent mollusc shells. Among the tested methods, specific staining with dyes against the DNA (nuclei) of the trace making organisms turned out to be a useful and reproducible approach. Bright and clearly delineated fluorescence signals of microendolithic nuclei allow, for instance, a differentiation between abandoned and still populated microborings. Furthermore, infiltrating the microborings with fluorescently stained resin seems to be of great capability for the visualisation and quantification of microbioerosion structures in their original spatial orientation. Potential fields of application are rapid assessments of endolithic bio- and ichnodiversity and the quantification of the impact of microendoliths on the overall calcium carbonate turnover. The method can be applied after CLSM of the stained microendoliths and retains the opportunity for a subsequent investigation of epoxy casts with SEM. This allows a three-fold approach in studying microendoliths in the context of their microborings, thereby fostering the integration of biological and ichnological aspects of microbial bioerosion.
The borings of microendoliths infiltrated with fluorescent resin were scanned and documented with the CLSM as image stacks (xyz-mode, see above) and subsequently loaded into Amira-Avizo Software Version 6.5. The segmentation editor and different marking tools of Amira such as the magic wand or the brush in combination with the ‘enable masking’ function have been used for a semi-automated segmentation of fluorescence signals of three different types of microborings inside a selected shell. Each ichnotaxon was assigned to a certain ‘material’ to calculate triangulated surface models (surface rendering). Different display and measuring modules of Amira were used for visualisation and quantification of the dataset.
For Research Use Only. Not for use in diagnostic procedures.